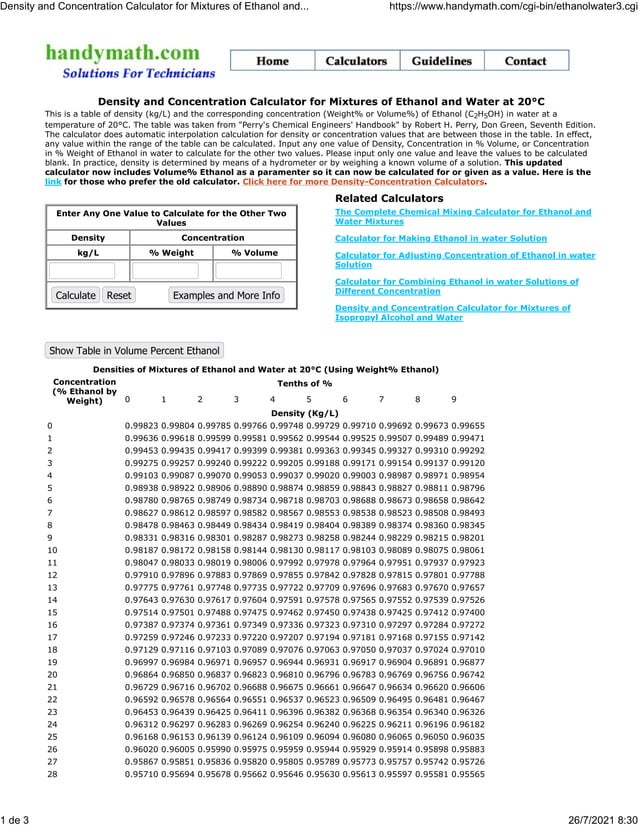
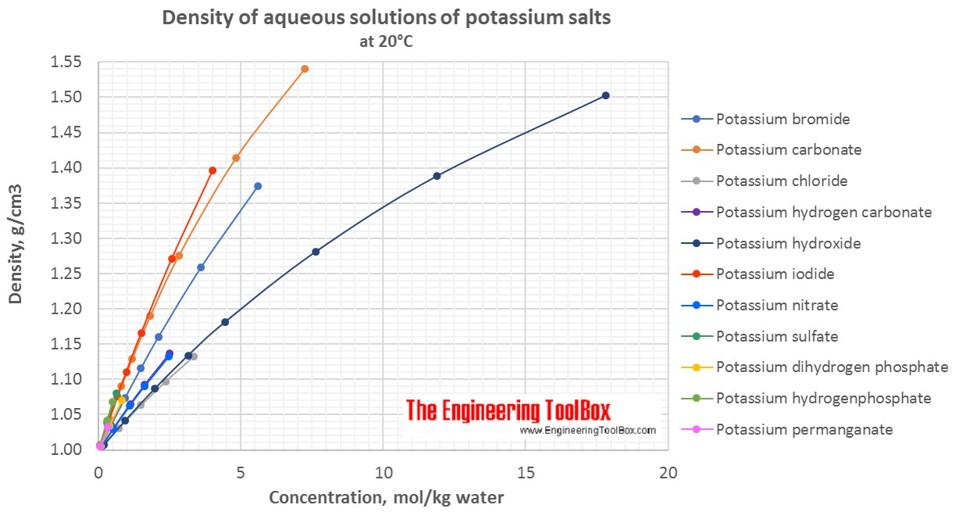
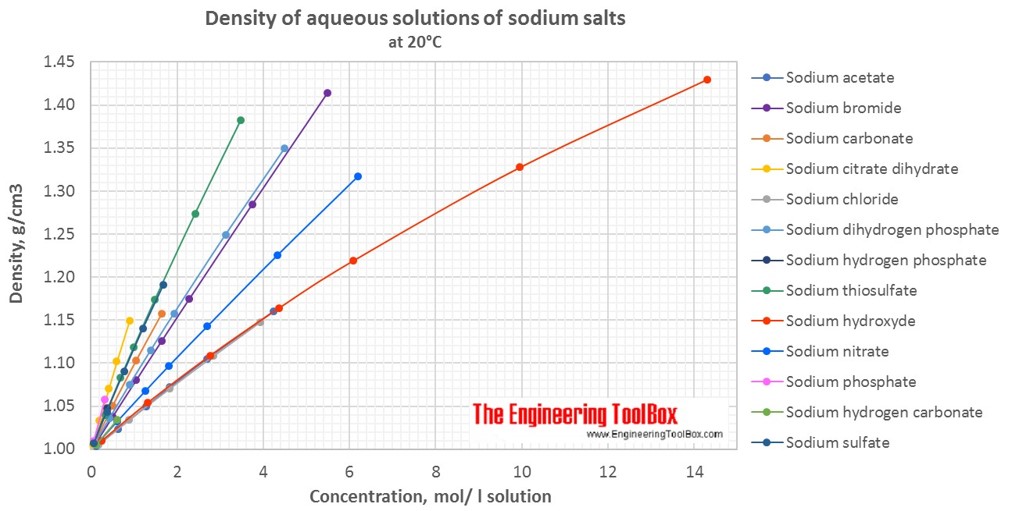
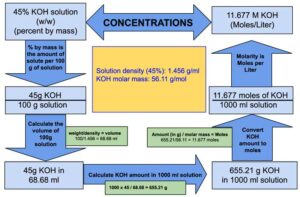
Calculate the molarity of `KOH` in solution prepared by dissolving `5.6 g` in enough water to - YouTube

A 7.0 M solution of KOH in water contains 28% by mass of KOH. What is density of solution in gm/ml ?

SOLVED: A 2.85 L aqueous solution of KOH contains 171 g of KOH . The solution has a density of 1.29 g/mL . Calculate the molarity ( 𝑀 ), molality ( 𝑚 ),

SOLVED: 114 Write the balanced chemical equation for the reaction of malonic acid with pO- tassium hydroxide: Calculate the molar concentration of the malonic acid if 38.17 mL of 0.315 M potassium

SOLVED: 5) Using the following data of the results of a titration with potassium hydroxide (KOH), calculate the concentration of sulfuric acid (HzSO4) Burette reading from the titration of 20.0 ml of